|
Why does Epsom salt make crystal spikes?
When you add Epsom salt to water, the salt dissolves. When
you leave the pan in the sun, the water evaporates and the salt forms crystals
shaped like long needles.
If you tried this experiment with table salt instead of
Epsom salt, you wouldn't get crystal spikes. That's because table salt and
Epsom salt are chemically different, so the crystals that they form are
very different.
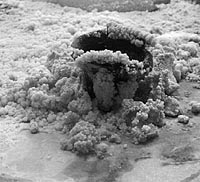
The picture on the right shows part of an artwork created
for the Exploratorium by Swiss artist Jörg Lenzlinger. He mixed different
kinds of salts with water. As the water evaporated, the salts crystallized,
making beautiful shapes that kept growing and changing.
To learn more about this salt crystal artwork, visit our
Complexity
website.
|